QUICK FACTS
Industry Engagement
MNSc Collaborations
SME Collaborations
Co-funding given to industries
Raised for Irish industries

Industry Representative Group
Roles & Function
The functions of the group include, but are not limited to, the following:
- Representatives from partner industries lead the Industry Representative Group (IRG), a forum that allows an exchange of ideas between partners and stakeholders.
- The IRG feedback enables the Centre to improve interactions with industry and support projects.
- It provides an opportunity to build relationships with new industries.
Expertise
Meet the
Team
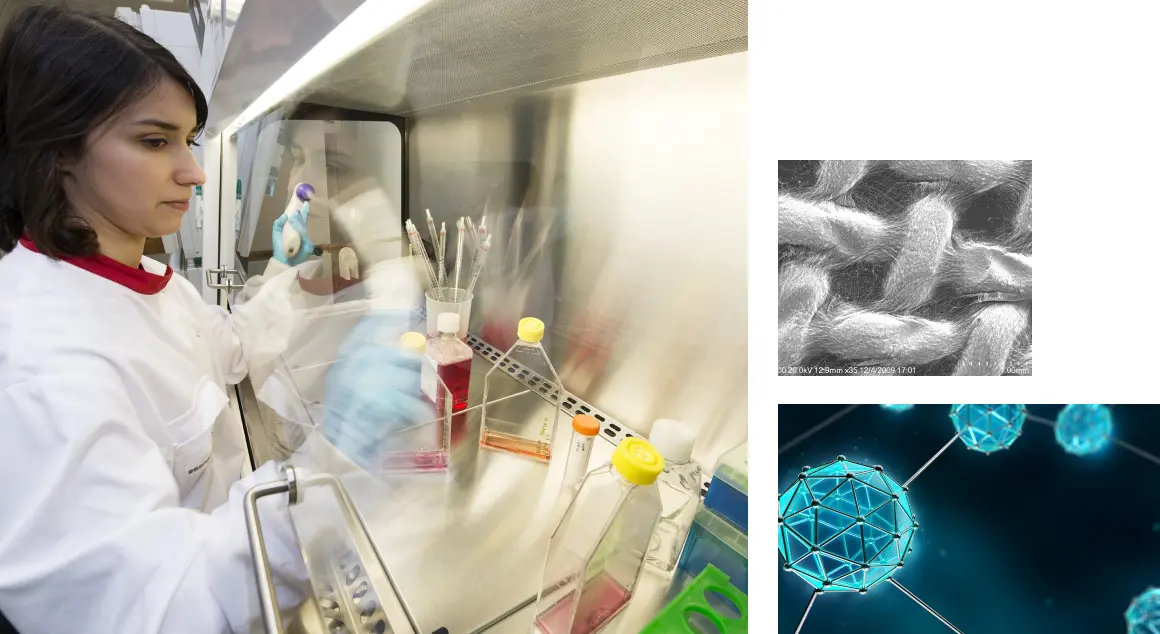
CÚRAM specialises in pioneering research across key areas such as biomaterials, drug delivery systems, tissue engineering, regenerative medicine, and medical device design. By collaborating with leading experts and leveraging cutting-edge technology, CÚRAM is dedicated to addressing the complex challenges of chronic health conditions like cardiovascular, musculoskeletal, neural, soft tissue, renal, urology, and respiratory diseases. Our ultimate goal is to translate innovative research into affordable, transformative medical solutions that improve patient outcomes and quality of life worldwide.
Technology Flyers
CÚRAM drives the translation of research into next-generation medical devices and implants. A collaborative approach combines fundamental and applied science with clinical and industry expertise. This booklet presents a selection of the innovative technologies being developed in the centre.